Research Shows that Black Plastic Contains Harmful Flame Retardant
 During recycling, according to a recent study, toxic flame retardants used to prevent fires in electronics are getting into the black-colored plastic that’s commonly used in takeout containers, children’s toys, kitchen utensils and grocery meat and produce trays. The most dangerous flame retardants were the ones used in the electronic cases of electronic devices like televisions. These items may be contaminated with worrisome amounts of toxic flame retardants, which have serious adverse health effects. If you or your child was harmed by black-colored plastic, you should call the seasoned Chicago-based product liability attorneys of Moll Law Group about whether you have a viable claim. Billions have been recovered in cases with which we’ve been involved. We represent consumers nationwide.
During recycling, according to a recent study, toxic flame retardants used to prevent fires in electronics are getting into the black-colored plastic that’s commonly used in takeout containers, children’s toys, kitchen utensils and grocery meat and produce trays. The most dangerous flame retardants were the ones used in the electronic cases of electronic devices like televisions. These items may be contaminated with worrisome amounts of toxic flame retardants, which have serious adverse health effects. If you or your child was harmed by black-colored plastic, you should call the seasoned Chicago-based product liability attorneys of Moll Law Group about whether you have a viable claim. Billions have been recovered in cases with which we’ve been involved. We represent consumers nationwide.
Contact Moll Law Group About Your Claim
The research study, conducted by Toxic-Free and the Amsterdam Institute for Life and Environment at Vrije Universiteit in Amsterdam, was recently published in the journal Chemosphere. It examined black plastics, but the researchers didn’t specify the brand names of what was tested. However there have been studies in other countries that similarly demonstrated that black plastic was contaminated with flame retardants.
It’s common for people to own electronic devices like gaming systems, TVs, smartphones, computers and tablets that include flame retardants in order to prevent fires. Flame retardants are also used in chairs, car upholstery, yoga mats, and padded baby items, and they can spread into the air and attach to food and water, which might then be consumed, or dust. When these items are put into recycling, they can contaminate other things
 Illinois Injury and Mass Tort Lawyer Blog
Illinois Injury and Mass Tort Lawyer Blog


 Even at low levels, benzene is a carcinogen and can increase the risk for blood tissue cancers like lymphoma, myeloma, and leukemia. Benzene can’t exist at any safe level in sunscreen, according to experts. Recently, Johnson & Johnson recalled certain Neutrogena and Aveeno products, because it was found that they contained benzene. This is not the first question about benzene in Johnson & Johnson sunscreens; in 2021, the
Even at low levels, benzene is a carcinogen and can increase the risk for blood tissue cancers like lymphoma, myeloma, and leukemia. Benzene can’t exist at any safe level in sunscreen, according to experts. Recently, Johnson & Johnson recalled certain Neutrogena and Aveeno products, because it was found that they contained benzene. This is not the first question about benzene in Johnson & Johnson sunscreens; in 2021, the  Alarmingly, the carcinogen glyphosate weedkiller has been found to exist in 60% of beans and lentils samples tested by labs working for the
Alarmingly, the carcinogen glyphosate weedkiller has been found to exist in 60% of beans and lentils samples tested by labs working for the  There have been a surge of shootings that may be the result of a defect in Sig Sauer P320. These incidents have happened unexpectedly and caused pain and devastation. For instance, a military sergeant was talking to his supervisor, when another soldier tried to get past him. The sergeant and soldier’s gun holsters clanked against each other, and the
There have been a surge of shootings that may be the result of a defect in Sig Sauer P320. These incidents have happened unexpectedly and caused pain and devastation. For instance, a military sergeant was talking to his supervisor, when another soldier tried to get past him. The sergeant and soldier’s gun holsters clanked against each other, and the  The average person who menstruates spends around five years of their lifespan using around 11,000 menstrual products, including tampons. A new study led by Jenni A. Shearston a postdoctoral scholar from UC Berkeley found that tampons from several brands that millions use can be
The average person who menstruates spends around five years of their lifespan using around 11,000 menstrual products, including tampons. A new study led by Jenni A. Shearston a postdoctoral scholar from UC Berkeley found that tampons from several brands that millions use can be  The Consumer Product Safety Commission has issued a
The Consumer Product Safety Commission has issued a  On May 30, 2024, the manufacturer Black & Decker
On May 30, 2024, the manufacturer Black & Decker  Recently around 1.6 million Black+Decker garment steamers were
Recently around 1.6 million Black+Decker garment steamers were 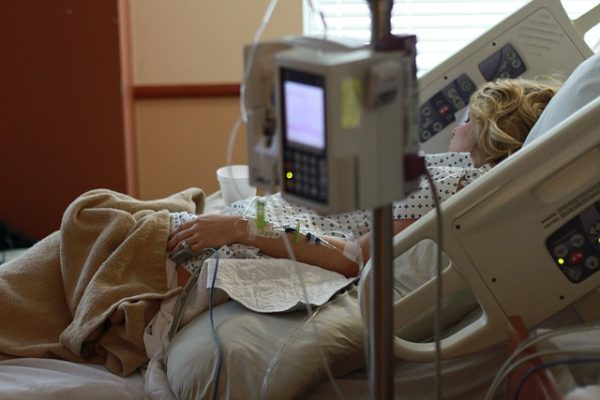 Around 15 million Philips Respironics CPAP and ventilator machines were impacted by a Class 1 recall. This class of recall is the most serious because it indicates that it’s reasonably probable that using or just being exposed to the affected products will result in serious adverse health ramifications or even deaths. Recently, the manufacturer Philips reached a
Around 15 million Philips Respironics CPAP and ventilator machines were impacted by a Class 1 recall. This class of recall is the most serious because it indicates that it’s reasonably probable that using or just being exposed to the affected products will result in serious adverse health ramifications or even deaths. Recently, the manufacturer Philips reached a  Doctors sometimes perform total hip arthroplasty or total hip replacement using the Synovo Total Hip System, which features resurfacing implants. The United States Food and Drug Administration (FDA) has
Doctors sometimes perform total hip arthroplasty or total hip replacement using the Synovo Total Hip System, which features resurfacing implants. The United States Food and Drug Administration (FDA) has